Rising ETO Incidents in the Global Spice Trade: A Technical Guide for QC and HACCP Teams
Ethylene oxide contamination continues to disrupt spice supply chains. Updated limits, recent recalls, and actionable HACCP strategies across major markets.

China Business Limited does not use ethylene oxide treatment on any products it sources or brokers, and does not work with suppliers who apply ETO treatment. We publish this guide because ETO contamination is a live risk in the broader spice trade — particularly for dehydrated garlic and cassia/cinnamon — and our buyers need to understand the regulatory landscape and testing requirements in their markets. If you are sourcing from suppliers outside our network, use this as a reference for what to demand.
Ethylene oxide (ETO) contamination in spices continues to disrupt global supply chains, with tightened regulations and recalls intensifying scrutiny. Following Hong Kong's June 2025 alert on Cape Foods black pepper, this updated guide provides Quality Control and HACCP teams with detailed technical data and actionable strategies to ensure compliance across major markets.
Latest Incident: Cape Foods Black Pepper (Hong Kong, June 2025)
On 17 June 2025, Hong Kong's Centre for Food Safety issued a recall for a 170g pack of "I Love Black Pepper with Grinders" (South African origin) after detecting ETO residues at 0.02 mg/kg — exceeding Hong Kong's zero-tolerance limit. The affected batch was traced to inadequate post-fumigation aeration at a shared processing facility. Sales were halted and a recall initiated.
ETO Regulatory Limits by Market
| Market | ETO Limit | Regulatory Reference | Notes |
|---|---|---|---|
| European Union | 0.1 mg/kg | Regulation (EC) No 396/2005 | Strictly enforced; RASFF alerts frequent |
| United Kingdom | 0.1 mg/kg | UK MRL legislation (retained EU) | Aligned with EU post-Brexit |
| United States | 0.1 mg/kg | 21 CFR Part 117 | FDA enforcement; state-level variation possible |
| Canada | 0.1 mg/kg | CFIA regulations | Nationwide recalls enforced for non-compliance |
| Singapore | 50 mg/kg (product-specific) | SFA regulations | Higher limit but zero for some categories |
| Hong Kong SAR | Zero tolerance | CFS / Cap. 132 | Any detectable level triggers recall |
| Australia | Zero tolerance | FSANZ Food Standards Code | Any detectable level triggers recall |
Compliance strategy: Align production to the strictest markets (Australia / Hong Kong: zero tolerance) with a single SOP. This eliminates the need for separate protocols per destination and reduces recall risk across all markets simultaneously.
Recent Global ETO Incidents
FDA recall of 500 kg bulk turmeric powder in Massachusetts — ETO at 0.15 mg/kg (limit 0.1 mg/kg). Contamination stemmed from over-fumigation in a multi-product facility.
RASFF alert for Indian cumin seeds — ETO at 0.18 mg/kg (limit 0.1 mg/kg). Shipments redirected for retesting; 30% failed secondary analysis.
FSA notice on Turkish chilli flakes — ETO at 0.12 mg/kg (limit 0.1 mg/kg). Retailers withdrew batches citing inadequate supplier aeration logs.
FSANZ recall of coriander seed pods — ETO at 0.09 mg/kg, violating Australia's zero-tolerance policy. Highlighted cross-contamination in packaging.
CFIA alert on Ontario-sold mixed spice packets — ETO at 0.14 mg/kg (limit 0.1 mg/kg). Nationwide recall enforced due to incomplete supplier records.
Bans on Indian spice brands MDH and Everest after detecting ETO in spice blends at 0.3–0.5 mg/kg — violating Hong Kong's zero-tolerance policy and Singapore's product-specific limits.
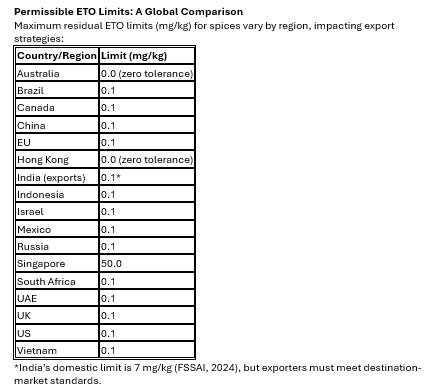 ETO regulatory limits across major export markets.
ETO regulatory limits across major export markets.
Understanding ETO: Technical Overview
What it is: Ethylene oxide (C₂H₄O, CAS 75-21-8) is a gaseous fumigant applied in sealed chambers (10–20 kPa, 40–50°C) to sterilise spices. Post-treatment aeration (25–30°C, 48–72 hours) is required to reduce residues.
Why it's used: Eliminates pathogens (Salmonella, E. coli) and moulds without thermal degradation of volatile compounds such as piperine in black pepper.
How contamination occurs:
- Inadequate aeration — Residual ETO binds to spice matrices, exceeding 0.1 mg/kg
- Over-fumigation — Excessive ETO (e.g. >500 ppm) due to uncalibrated equipment
- Cross-contamination — Shared fumigation chambers or packaging lines transferring residues
HACCP-Aligned Best Practices for ETO Control
Enhanced Testing
- GC-MS (LOD 0.01 mg/kg) at raw material intake, post-fumigation, and pre-shipment
- Third-party labs accredited to ISO/IEC 17025:2017
- Statistical sampling (AOAC binomial) for 95% confidence
Supplier Audits
- Audit fumigation facilities for equipment calibration and aeration protocols
- Verify CoAs and ETO application logs against batch records
- SOPs for chamber decontamination
Traceability
- Blockchain or QR-code lot-level tracking — isolate affected lots within 4 hours
- Integrate with ERP systems for real-time batch monitoring
- ISO 22000:2018 compliance
Alternative Sterilisation
- Steam: 120°C, 15 psi, 20 min — no chemical residues
- Irradiation: 5–10 kGy (requires labelling in EU, US)
- Ozone: 5–10 ppm, 30 min — emerging method
- Testing methods: Use GC-MS or LC-MS/MS with LOD ≤0.01 mg/kg. Validate against AOAC International standards (e.g. AOAC 2019.01). Headspace GC-MS achieves LOD of 0.005 mg/kg — preferred for zero-tolerance markets.
- CCPs: Establish Critical Control Points at raw material intake, fumigation, and packaging. Monitor ETO residues at each stage.
- Corrective actions: For non-compliant batches — re-aerate (72 hours, 25°C, 500 m³/h airflow) or divert to markets with higher limits.
- Risk assessment: Conduct hazard analysis for ETO as a chemical contaminant under Codex Alimentarius HACCP principles. Identify raw material sourcing and fumigation as high-risk stages.
- Documentation: Maintain CoAs for each batch specifying ETO testing method, LOD, and compliance with destination market regulations.
- Segregation: Store and process ETO-treated and non-treated spices separately to prevent cross-contamination.
- Training: Annual training of QC staff on ETO testing methods and HACCP principles per ISO 22000:2018.
- Regulatory monitoring: Subscribe to real-time alerts from FDA, EU RASFF, UK FSA, FSANZ, and CFIA. The EU and US are exploring lower limits of 0.05 mg/kg by 2027.
- Continuous improvement: Use Plan-Do-Check-Act (PDCA) cycles to refine ETO controls, integrating feedback from regulatory rejections.
- CFS (2025). Food Safety Alert: Black Pepper Recall. Hong Kong Centre for Food Safety.
- Codex Alimentarius (2020). HACCP Guidelines.
- EFSA (2025). Predictive Analytics for Food Safety.
- FAO (2025). Global Spice Trade Report.
- FSSAI (2024). Spice Export Regulations.
- ISO 11135:2014. Sterilization of Health Care Products.
- ISO 22000:2018. Food Safety Management Systems.
- AOAC 2019.01. Method Validation Standards.
This post is intended for informational purposes only. Regulatory limits and enforcement practices change frequently — always verify with official government sources or accredited laboratories before making compliance decisions. China Business Limited accepts no liability arising from use of this information.
Sourcing spices and ingredients that are ETO-free by design? That's our starting point, not an afterthought.
Talk to Us
